In October we hosted another hugely successful User Symposium to give our customers a chance to connect and share their experiences. This year we were joined virtually by 245 attendees from 24 countries. The live sessions were recorded and accessed on-demand until the 12th of November.
During the Symposium, Dotmatics scientists provided updates on development Roadmaps covering all strategic areas: Biologics Discovery, Small Molecule Discovery, Chemicals & Materials Innovation and Lab Data Automation. We were joined by experts from Debiopharm, Charles River Laboratories, Casma Therapeutics, Apollo Therapeutics,Firmenich S.A, and LabVoice who shared their experiences of using Dotmatics software to streamline their scientific projects.
The annual User Symposium is one of the highlights of our events calendar and we always look forward to connecting with customers and providing a platform to exchange experiences in a supportive environment. Here are what some of the attendees thought about this year’s symposium:
“I’m always keen to find out what’s coming up in new products that’s why I always make sure to attend this event.”
“Great to hear how others are implementing the Dotmatics system.”
“Important to keep up to date with the up and coming developments at Dotmatics.”
“Impressive speaker line-up with useful contributions.”
“Looking forward to next year’s symposium!”
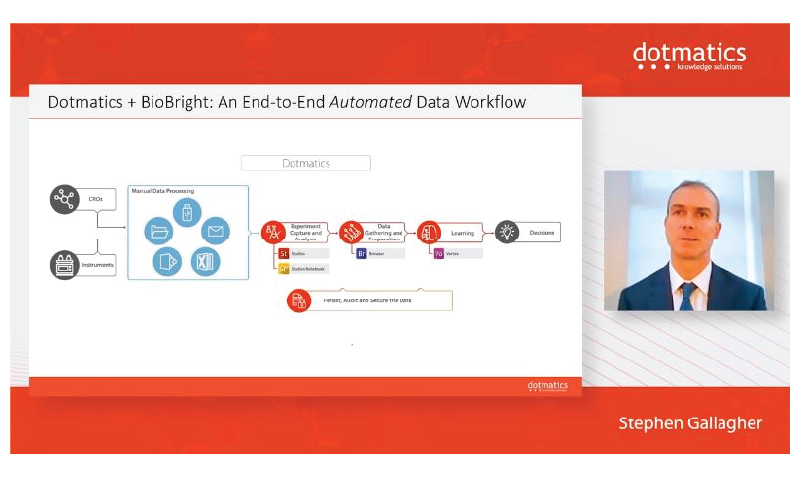
The event was opened by Dotmatics CEO Steve Gallagher and, in addition to the Live Stream, educational sessions ranging from tips & tricks, new biologics tools, implementation of AI, parallel chemistry workflows, latest enhancements to Screening Ultra to BioBright & Virtru were accessed on-demand.
Next up, we invited speakers from the scientific community to share their experiences of using Dotmatics in their organizations. here are some highlights from their talks:
Jean Richardson, IT Scientist – Debiopharm “Running a Tight Ship”.
Jean outlined the complexities of sample shipping and management when working with a network of CRO synthesis and analysis partners. Debiopharm, a Swiss biopharmaceutical company with a focus on oncology and bacterial infection therapeutic areas, required accurate tracking of shipped samples along with accurate and reliable measurements of available stock. Debiopharm scientists now have a streamlined shipping request workflow allowing them to focus on the scientific aspects of projects rather than logistics.
Riannon Hambleton, Associate Director, Business Technology Partnership – Charles River Laboratories “If you give a Scientist an Electronic Lab Notebook…”.
Riannon shared the Charles River Labs experience in implementing an ELN. CRL have sites all over the world in a variety of areas including discovery, research models, safety assessments, biologics testing and microbial QC. The roll out of an ELN in Discovery is part of a strategic move to use the CRL global scale to streamline and improve processes with an emphasis on cloud and SaaS capability. Riannon shared how to keep the project manageable and deliver value early to the scientists. Notably, CRL embraced video-based training as part of go-live and Riannon’s project implementation team have had very positive feedback on the overall experience.
Jarrod Medeiros | Director, Informatics and IT – Casma Therapeutics “Beyond ELN: The Dotmatics Platform at Casma”.
Casma therapeutics successfully deployed an ELN for their biologists during the pandmeic lockdown in springl. Jarrod outlined some follow-on projects which have been prioritised on business need basis. One of which was bringing compound management in-house and using the ELN to provide a workflow for sample request & fulfillment. Jarod also updated attendees on the Casma High Content Screening workflow project involving integration between BioBright and Dotmatics systems.
Charlotte Lane | Project and Operations Director – Apollo Therapeutics “Apollo Therapeutics: How we use Dotmatics”.
Charlotte shared her experince of using the Dotmatics platform over the past three years. Apollo Therapeutics is a collaborative venture between Imperial College London, University College London and the University of Cambridge and three global pharmaceutical companies AstraZeneca, GlaxoSmithKline and Johnson & Johnson Innovation. The company is indication and modality agnostic with an active portfolio of 22 projects. It’s a virtual organization working with over 15 different CROs for whom data security and integrity is key.
Marco Pacchiani | Vice President, Innovation Process & Knowledge Management – Firmenich S.A. “Towards a Data Driven Organization”.
Firmenich is a Swiss and family-owned for over 120 years, creating fragrances and flavors for the world’s most desirable brands. Marco provided context on the Dotmatics implementation project for knowledge generation capability within Firmenich corporate research which involved replacing an outdated legacy system with weak data features. Céline Amoreira, Senior Scientist, gave details of the project implementation.
Dan DeAlmeida | Director of Product Management – LabVoice.
Dan demonstrated integration between the Dotmatics ELN and LabVoice’s scientific vocabularies and voice transcription capabilities to streamline R&D data capture. Scientists can operate in a hands-free environment, improving workflow efficiency and data integrity.
The event rounded off with informal networking in a virtual lobby and “Technical Tent” hosted by Spatialchat – this proved a particular hit with everyone able to join in groups of chats. Not quite the live experience of a conference but a good second best!
We hope you enjoyed attending the 2020 Dotmatics User Symposium as much as we did hosting it. The success is largely due to end-user participation, from both attendees and speakers and we were fortunate to have thoughtful and engaging presentations from our guests yet again this year along with an abundance of enthusiastic questions from customers.
We’re already planning for next year and are hopeful that the global situation will allow us to host a combined in-person and streamed event resulting in the best of both!